Current collectors serve two purposes in lithium batteries: as conductors and as carriers for active materials. They conduct current and support the powder-like active materials, forming a complete system. Currently, aluminum foil is used for the positive electrode, and copper foil is used for the negative electrode because aluminum and copper can meet the requirements of conductivity and load-bearing.
The reason why both the positive and negative electrodes cannot use aluminum foil or copper foil is mainly due to the difference in discharge principles. If copper is used for the positive electrode, the lithium ions in the lithium battery will be directly consumed due to chemical reactions. Other metals (such as nickel-chromium) can also be used for both the positive and negative electrodes, but aluminum and copper are cheaper and have certain ductility, allowing for thinning. Therefore, for lithium battery systems, aluminum foil for the positive electrode and copper foil for the negative electrode are the best choices.
As a carrier material, flexibility is required. During battery production, the current collector serves as a substrate, and other materials need to be coated on it. This process requires stretching during production, and in the assembly process, it needs to be "rolled" and undergo bending. This requires the current collector material to have a certain level of flexibility. Otherwise, it may fracture at bent positions, leading to short circuits in the battery. Flexibility also affects battery life and cycle count.
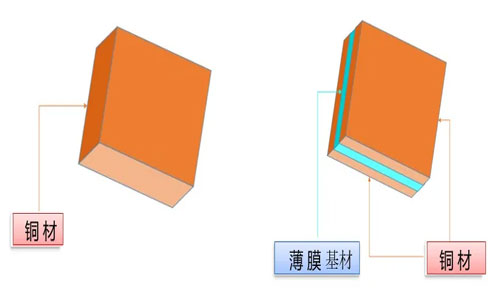
Composite copper foil is a new type of current collector material for dynamic batteries, mainly composed of three parts: a middle layer of PET, PP, PI, or other similar materials as the base film, and copper layers with a thickness of about 1μm on both sides. It is a sandwich-like structure.
The manufacturing process of composite copper foil is different from that of traditional metal foils. Traditional copper foil is mainly produced through rolling or electroplating processes, while composite copper foil is made by magnetron sputtering or vacuum deposition of a 20-80nm copper layer on the surface of a plastic film with a thickness of 3.5-6μm, and then electroplating to increase the thickness of the metal layer to 1μm, resulting in a composite metal foil with a total thickness between 5.5-8μm, replacing the 6-9μm electrolytic metal foil.
Compared to traditional metal current collectors, composite copper foil has the following characteristics:
[1] Good flexibility: The electrical conductivity of the PET substrate relies on the surface metal. The intermediate polymer material has better flexibility than metal. Composite current collectors, therefore, have better flexibility due to the intermediate properties. This enhanced flexibility makes them more practical.
[2] High compressive strength: This mainly measures tensile performance. During battery production, the current collector needs to undergo processes such as "winding, pulling, and laying." It needs to withstand greater tension during these processes. High polymer materials have better compressive strength than metals, so the tensile strength of composite materials is also better than pure metals. This means that the increased tensile strength allows for the use of greater tension in the production process, thereby improving production efficiency. Battery factories are currently expanding new production lines, and if PET materials are used, production can be accelerated on the existing production lines.
[3] Low density, low weight, and high gravimetric capacity: To increase the energy density of batteries, materials can either increase the energy directly or reduce the weight of the materials. The active materials in batteries are at the nanometer level, and thinning the current collector can release electrical energy more evenly. Approximately 1 GWh of batteries requires 10 million square meters of both positive and negative current collectors, so the proportional area is fixed. Currently, aluminum and copper are the best choices for lithium battery systems, and the materials are relatively fixed. The density cannot be changed, so the volume can only be reduced by reducing the thickness to reduce weight and achieve higher energy density. For example, contemporary battery manufacturers use 6μm copper foil, and even laboratory experiments use 3.5-4μm copper foil. However, copper foil cannot be thinned indefinitely, and the thinner the copper foil, the weaker its compression strength.
Taking 6μm PET copper foil as an example, the middle layer is a 4μm PET material, and there is a 1μm copper foil on each side, for a total thickness of 6μm. Two-thirds of the total thickness is pure copper replaced by PET, with the added benefit of PET having a much lower density (only 1/10 of copper's density). The density of the entire material can be reduced to 38.5%. Therefore, a 6μm PET current collector is equivalent to the weight of a 2.4μm copper foil. PET current collectors provide a method of increasing battery energy density without sacrificing load-bearing capacity, safety, and cycle life by overly thinning the collector.
[4] Good uniformity: During conventional production processes, two issues may arise. One is the uniformity of the double-sided properties of the film. From a process perspective, the binding agent of the active material needs to be used to adhere it to the surface of the PET film, followed by rolling to ensure the uniformity of the surface thickness and prevent unevenness that could result in material detachment. The other issue is the asymmetry of contact resistance. The resistance of the active material is different from that of copper, with some areas having more resistance and others having less. Such uneven energy distribution can affect overall discharge performance and, subsequently, cycle life, accelerating battery decay. Currently, copper foil treatment includes single-sided roughening, double-sided roughening, and surface roughening. This indicates that the two sides of copper foil are not equal and that there are issues with uniformity.
PET current collectors have addressed these issues. Firstly, PET material itself has relatively uniform properties on both sides. In addition, during the coating of the active material, physical deposition, electroplating, and other methods are mainly used to achieve a more uniform surface. Due to the uniformity of the PET surface, the lower expansion rate of the polymer material, and the ease of vacancy retention, the surface integrity is maintained, resulting in longer lifetimes.
[5] High safety: Battery combustion is caused by uncontrolled heat generation leading to internal short circuits. The battery structure consists of two metal poles and a separator in the middle. Metal materials, due to repeated use, can experience fatigue fracture, resulting in small spurs forming on the edges. These spurs can easily squeeze onto the separator, puncturing it. Joining the positive and negative poles together, combined with the release of active substances in the electrolyte which are in liquid form, and the jolting of the battery during electric vehicle use, internal short circuits can easily occur, releasing a large amount of heat. The solution provided by PET copper foil is that high polymers are not easily broken. Even if they break, the strength of the 1μm copper plating cannot meet the standard for puncturing the separator, effectively avoiding the risk of internal short circuits.
[6] Low cost: Composite copper foil is less sensitive to copper price fluctuations,
Contact: Pamela
Phone: +86 189 6365 3253
E-mail: info@industryprocess.com
Whatsapp:+86 189 6365 3253
Add: Yajing Industrial Park, No. 59 Shuangjing Street, Weiting Town, Suzhou Industrial Park
We chat